Chronic venous insufficiency
Understanding CVI
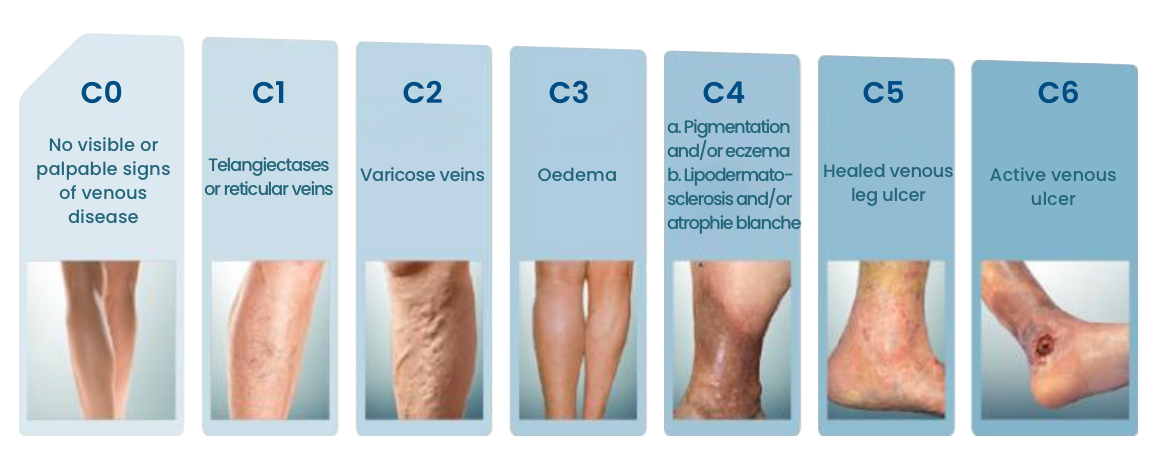
Disease Progression
If untreated, patients suffering from CVI disease can expect their condition to worsen over time
Prevalence of Venous Reflux Disease
An estimated 30-35 million people in the US have CVI of the legs¹ , making CVI 3 times
more prevalent than heart disease
Another European study estimates the probabilities for having CVI for
each gender and age group as follows² :
¹ Brand FN, Dannenberg AL, Abbott RD, KannelWB. The epidemiology of varicose veins: the Framingham Study. Am J PrevMed 1988; 4(2):96-101 Coon WW, Willis PW, Keller JB. Venous thromboembolism and other venous disease in the Tecumseh Community Health Study Circulation 1973; 48:839-846
² The influence of age and gender on venous symptomatology. An epidemiological survey in Belgium and Luxembourg, Show all authors Marc E Vuylsteke, Roos Colman, Sarah Thomis, Geneviève Guillaume, Evy Degrande, Ivan Staelens. First Published June 1, 2015
Target Market
1.7 Million Patients Seeking Treatment
Of these 30-35 million people, 1.7 million seek treatment annually. Of the 1.7 million people seeking treatment annually, approximately 700,000+ undergo endovenous vein ablation procedures to treat CVI that are covered by insurance:
These 700,000+ procedures in the USA are the procedures that we expect to displace with the Seclusion™ catheter and procedure.
Current Treatment Options
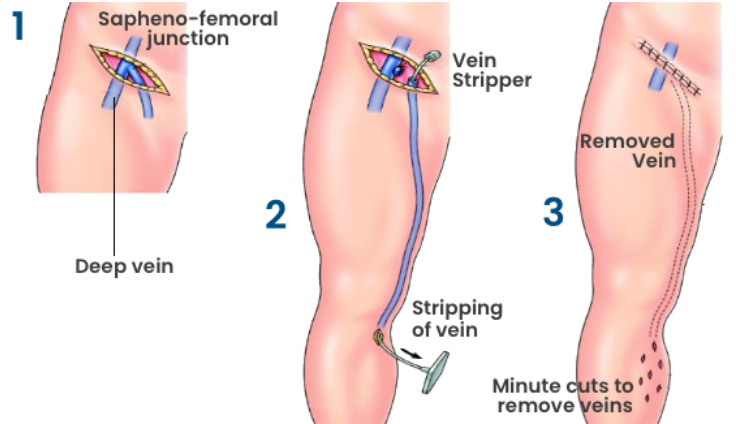
Vein Stripping
Vein stripping is a surgical procedure done under general or local anaesthetic to aid in the treatment of varicose veins and other manifestations of chronic venous disease. The vein “stripped” is usually the great saphenous vein.
Shortcomings
- Invasive: requires surgery and anesthesia
- Long recovery time
- High reoccurrence rate
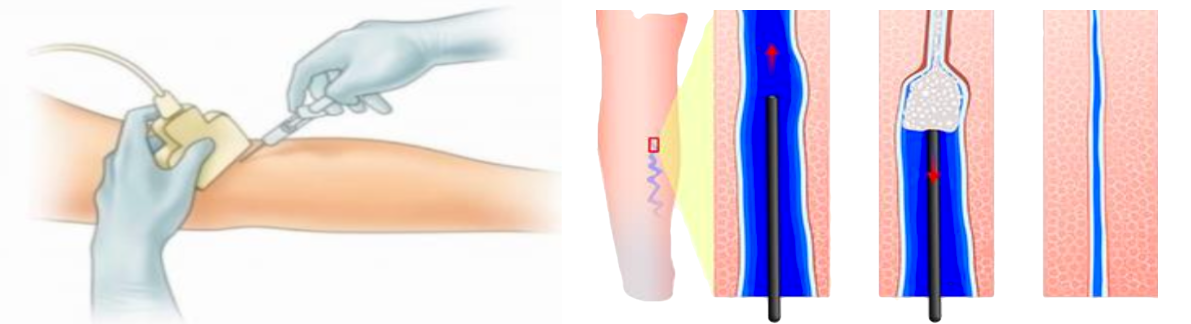
Ultrasound Guided Injection Sclerotherapy
Ultrasound Guided Injection Sclerotherapy is a commong procudure that is currently offered by many groups including Angiodynamics (Asclera®), BTG
International/Boston Scientific (Varithena®), as well hundreds of specialty pharmacies.
Shortcomings
- Sclerosant remains in the body
- Too much sclerosant has systemic toxicity issues
- Denoted headaches from migrating sclerosant
- Low efficacy treating larger veins
Endovenous Radio Frequency Vein Ablation
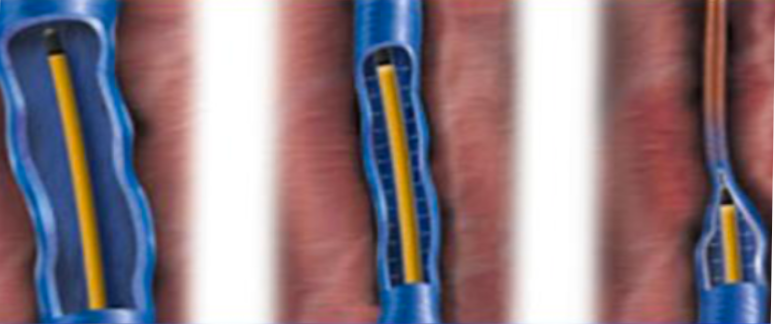
Endovenous Radio Frequency Vein Ablation is a treatment currently offered by Medtronic and marketed under the name ClosureFast™. It is currently regarded as the Gold Standard for CVI treatment.
Shortcomings
- Many painful needle sticks
- Unsafe to use below the knee
- Toxicity of tumescent anesthesia
- Tissue/nerve damage from heat (2500°F)
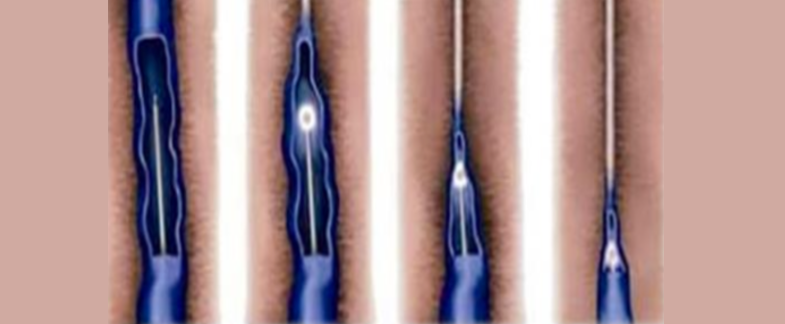
Endovenous Laser Vein Ablation
Endovenous Laser Vein Ablation is a treatment currently offered by many laser companies.
Shortcomings
- Many painful needle sticks
- Unsafe to use below the knee
- Toxicity of tumescent anesthesia
- Tissue/nerve damage from heat (600°F)
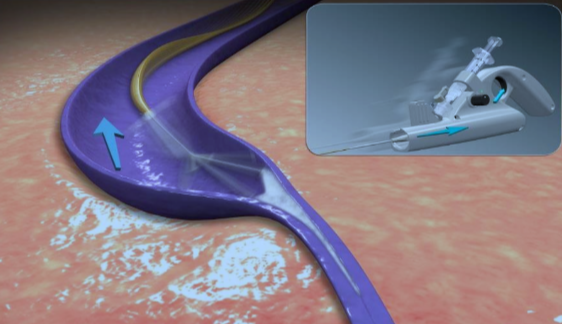
ClariVein®
ClariVein®IC is a specialty infusion catheter with a rotating wire tip designed for the controlled 360-degree dispersion of physician-specified agents to the targeted treatment area.
Shortcomings
- Sclerosant remains in the body and is toxic
- Questionable effectiveness
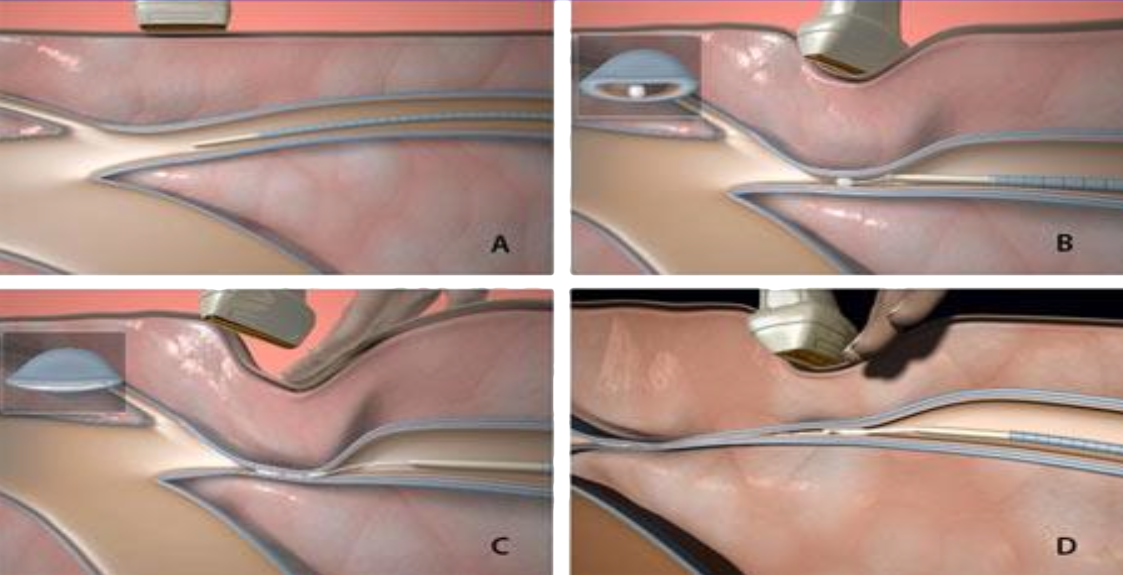
VenaSeal™
VenaSeal™ is a treatment currently offered by Medtronic that is glue-based and relies on permanent implantation.
Shortcomings
- The “Super Glue” is a permanent implant
- Foreign body (implant) reaction is unknown
- Arduous procedure
The Competition
While there is competition in the space, it is fractured across the different treatment types
There are over 5,500 hospitals and vein centers currently performing vein ablations in the US. Of those we want to focus on those doing over 40 Vein Ablations / month, leaving us a target market for commercialization of 1,900 centers.

