Vein Ablation SOLUTIONS
The Seclusion™ Catheter
We’re proud to introduce the first ever catheter that meets all the needs of vein ablation therapy. It’s easy, it works, and it’s going to change the way you treat patients and do business.
What Is BASIS™?
BASIS™ in an acronym that stands for Balloon Augmented Segmental Intraluminal Seclusion
Basically, it’s an endovenous, chemical ablation procedure for the treatment of superficial venous insufficiency, which uses a dual-balloon system (7 cm apart). These two balloons are inflated to isolate (seclude) a specific segment of a diseased blood vessel, remove the blood therein, infuse an FDA approved ablative Agent (sclerosant) for a specified period of time, then remove the sclerosant, and finally, deflate the balloons and reposition the catheter for the next treatment segment of diseased vein. Only 1 needle is required to gain access to the diseased vein.
device specifications
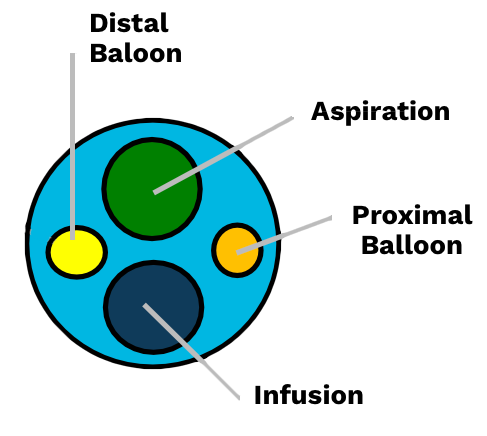
Anatomy of The Seclusion™ Catheter
- P 85cm Length, 7fr
- P7 and 3.5cm Marker Bands
- P4 Lumens
- P2 Balloons, 7cm Apart (12mm Diameter)
- P3 Aspiration and 4 Infusion Openings
- PHand Controller
- PAccommodates an 0.014 or 0.018 Guidewire

Intellectual Property
BASIS™ Patent Filings
To date, Basis has been granted 5 Patents. A complete list of published and pending patent applications can be seen on the following page.
- U.S. Patent No. 11,083,463
- U.S. Patent No. 11,166,729
- U.S. Patent No. 11,571,217
- U.S. Patent No. 11,666,744
- CA Patent No. 2,986,475
Endovenous Vein Ablation Market Need
Physicians that provide vein care have been asking for a pain- free, safe, effective, non-thermal, and non-tumescent vein ablation device for the treatment of their patients suffering from chronic venous insufficiency disease of the leg. Patients have also been asking for a less painful procedure.
The Seclusion™ Catheter will be the first such device offered to meet these market needs.
The Clinical Value of Seclusion™ Over Current Therapies
- Non-Thermal
- No Tumescent
- No Painful Injections
- No Lidocaine Toxicity
- No Incidental Tissue or Nerve Damage
- Direct Vein Wall Contact with Non Diluted Agent
- Projecting Excellent Long-Term Clinical Efficacy
- Targeted Precise Placement of Agent
- Removal of Agent after Treatment
- No Agent Migration (Secluded Treatment Area)
- No Agent/Sclerosant Toxicity Issues
- Not an Implant. No Painful Foreign Body (Implant) Reaction
- No Migration (of Implant) Risk
- No Intra-Operative External Groin Pressure Needed
- Physician has the Option to Treat a Maximum of 20 7cm Vein Segments in One Visit